There are essentially three main methods used to decaffeinate coffee; water-processed, chemical solvent processed, and the most recently devised method is the C02 process. Below are brief descriptions of each.
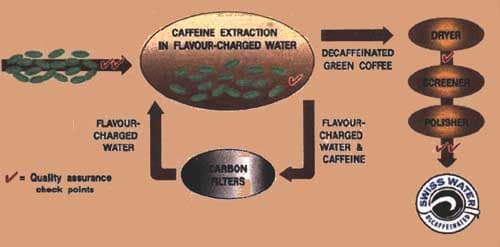
Water Processed. The most common of water-processed coffee decaffeination is the patented Swiss Water Process. There are others such as Royal Select Water process. Both are similar in that no chemicals are used to decaffeinate the coffee beans.
Raw beans (green coffee) are placed under pressurized, heated water which circulates through them causing the beans to absorb the water and swell. The pores of the coffee beans open, allowing the caffeine to wash free of them. The now resulting aquafied, caffeinated substance is run though a series of activated carbon filters, thus indirectly removing the caffeine from the coffee beans. The remaining substance is flavor-charged water (without caffeine), and is circulated back into the beans which adds the flavor back into the coffee. Next, the beans are dried, polished, bagged and made ready for shipment to the roaster.
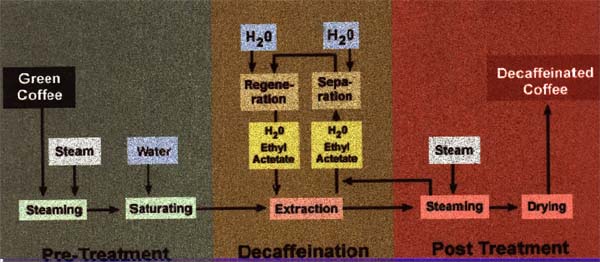
Solvent Method – (methylene chloride and ethyl acetate) – Okay, this sounds a lot scarier than it is. While neither of these methods has been labeled unhealthy by the medical establishment, Methylene chloride is a chemical that has been implicated in the depletion of the Earth’s ozone layer. Ethyl acetate is found naturally in fruit, and such you may see coffee by this process called natural processed or naturally decaffeinated. Keep in mind that both of these chemicals are very volatile (they evaporate easily) and therefore it is very unlikely that any residues survive the high temperatures of the roasting and brewing processes that occur before the coffee is actually consumed. Nevertheless, consumers concerns and fears of such substances have led to the development of the other processes. The solvent method is also called such names as European Process, Traditional Process, and Conventional Process.
The process itself goes something like this: Moistened coffee beans are soaked in water and pressurized in a steam environment. The beans swell making the caffeine accessible to extraction and are then placed in contact with the chemical. Once the desired decaffeination level is achieved (typically 99% or better), the beans are removed, dried, and are ready for roasting.
Carbon Dioxide or C02 Process – A vital element and important compound of our natural environment is carbon dioxide. It is in the air we breath, it is also the gas that makes mineral water effervescent, and by assimilation it enables plants to grow. It is also a highly selective solvent for caffeine. In this patented process, the natural carbon dioxide is used under sub-critical conditions (in a liquid state at low temperature and pressure, relative to the super-critical process). These process parameters, together with the exceptional caffeine selectivity of CO2, make for a high retention rate of coffee components responsible for aroma and taste. The process can be described as follows:
Green coffee beans are moistened with water and put into a vessel where it is in contact with pressurized liquid carbon dioxide. By circulation through the coffee, the carbon dioxide draws the caffeine out of the beans. In an evaporator the caffeine precipitates from the CO2, which after evaporation and re-condensation is pumped again into the coffee-containing vessel for a new cycle. When the required residual caffeine level is reached, the CO2 circulation is cut short and the coffee is discharged into a drier where it is gently dried to about the original moisture content. After that the coffee is ready for roasting.
Some specific characteristics of the Natural Liquid Carbon Dioxide Coffee Decaffeination Process are:
- The compounds responsible for flavor and taste development in the roasted coffee are practically left intact (this is especially beneficial for high-grade coffees).
- Certification as “organic” and “Kosher” by appropriate organizations/authorities.
- Shelf life is about the same as regular “caffeinated” green coffee beans.
Happy Brewing!
